 
The properties and the processing of ceramics are largely affected by their grain sizes and shapes, and characteristics such as density, hardness, mechanical strength, and optical properties strongly correlate with the microstructure of the sintered piece. Due to these differences (e.g., multiple crystals with various orientations, presence of grain boundaries, fabrication processes), polycrystalline materials should really not be confused with single crystals and should be the only ones included under the definition of ceramics. Fabrication processes of polycrystalline materials are relatively inexpensive, when compared to single crystals. Polycrystalline materials are formed by multiple crystal grains joined together during the production process, whereas monocrystalline materials are grown as one three-dimensional crystal. Sometimes, even monocrystalline materials, such as diamond and sapphire, are erroneously included under the term ceramics. Ceramics are by definition natural or synthetic inorganic, non-metallic, polycrystalline materials. The properties of ceramics, however, also depend on their microstructure. The ionic and covalent bonds of ceramics are responsible for many unique properties of these materials, such as high hardness, high melting points, low thermal expansion, and good chemical resistance, but also for some undesirable characteristics, foremost being brittleness, which leads to fractures unless the material is toughened by reinforcing agents or by other means. For this reason, polymers are very elastic (e.g., a rubber band), can be easily melted, and have low strength. Of all the four types of bonds, Van der Waals is the weakest. In polymers, there are covalent bonds between the atoms of the polymer, but the polymeric macromolecules (or chains) are kept together by Van der Waals forces. 
An example of Van der Waal bond is the hydrogen bond between hydrogen and oxygen, which is responsible for many properties of water. Van der Waals bonds consist of weak electrostatic forces between atoms that have permanent or induced polarization. The free movement of electrons also explains why metals tend to be conductors of electricity and heat. Metallic bonds are responsible for the main properties of metals, such as ductility, where the metal can be easily bent or stretched without breaking, allowing it to be drawn into wire. Metallic bonds are not as strong as ionic and covalent bonds. In the first one, the metal cations are surrounded by electrons that can move freely between atoms. There are two other types of atomic bonds: metallic and the Van der Waals. The two ions having opposite charges attract each other with a strong electrostatic force.Ĭovalent bonding instead occurs between two nonmetals, in other words two atoms that have similar electronegativity, and involves the sharing of electron pairs between the two atoms.Īlthough both types of bonds occur between atoms in ceramic materials, in most of them (particularly the oxides) the ionic bond is predominant. In an ionic bond, one of the atoms (the metal) transfers electrons to the other atom (the nonmetal), thus becoming positively charged (cation), whereas the nonmetal becomes negatively charged (anion). 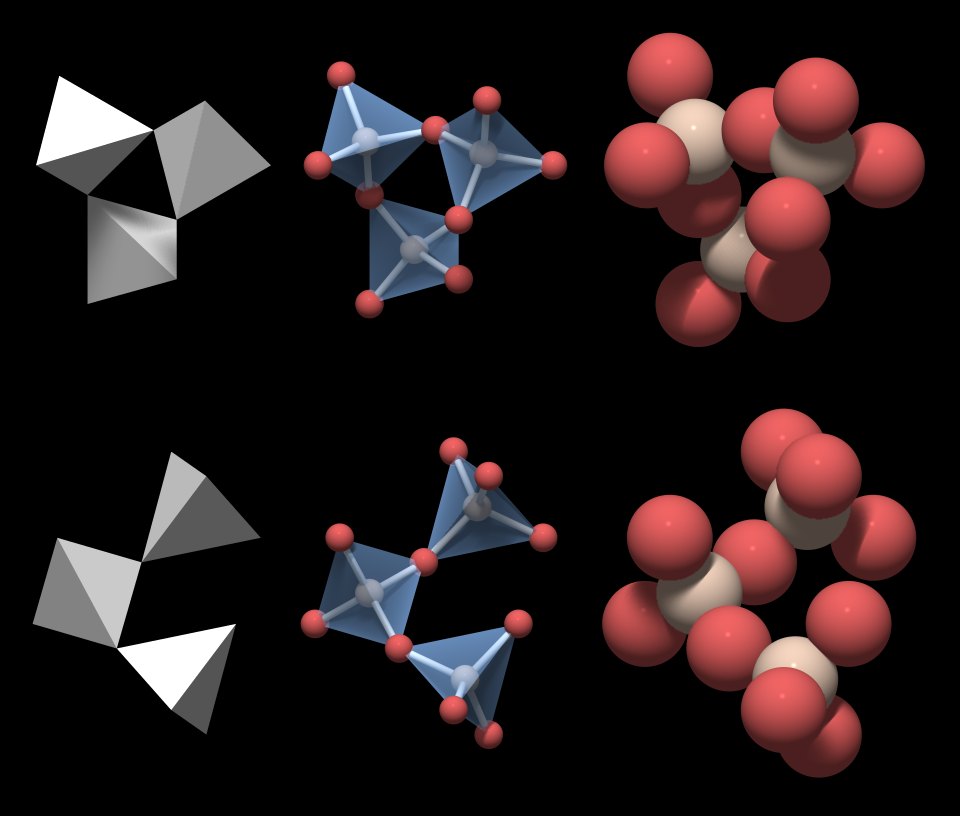
Electronegativity is the capability of the nucleus in an atom to attract and retain all the electrons within the atom itself, and depends on the number of electrons and the distance of the electrons in the outer shells from the nucleus. The ionic bond occurs between a metal and a nonmetal, in other words, two elements with very different electronegativity. Two types of bonds are found in ceramics: ionic and covalent. Arrange the solids in order of increasing melting points based on your classification, beginning with molecular solids.Ī.Just like in every material, the properties of ceramics are determined by the types of atoms present, the types of bonding between the atoms, and the way the atoms are packed together.Based on their positions, predict whether each solid is ionic, molecular, covalent, or metallic. Locate the component element(s) in the periodic table.\) as ionic, molecular, covalent, or metallic solids and arrange them in order of increasing melting points.Īsked for: classification and order of melting points 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
